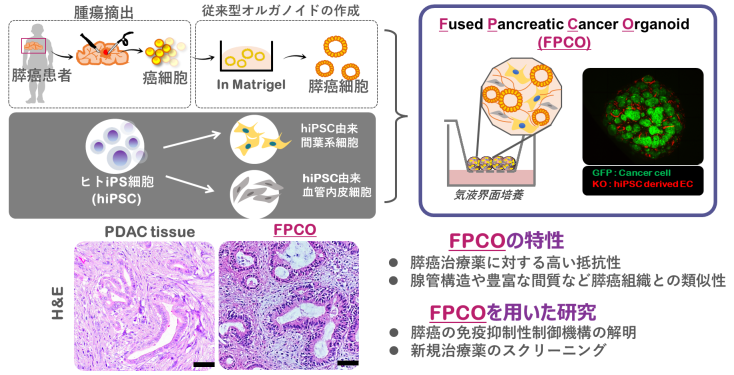
ヒトiPS細胞を活用した癌微小環境を再現する膵癌オルガノイドの創出
膵癌(主にPancreatic ductal adenocarcinoma, 以下PDAC)は5年生存率10%未満の予後不良な難治性の癌であり、未だ有効な治療法が存在しない。近年、免疫細胞や腫瘍血管、線維芽細胞、さらに豊富な細胞外基質から構成される膵癌に特徴的な微小環境と予後不良の要因が関連付けられるようになっている。我々は、膵癌手術検体から得たプライマリヒト膵癌細胞およびヒトiPS細胞由来間葉系細胞・血管内皮細胞を用いて、膵癌微小環境を再現した膵癌オルガノイド(Fused Pancreatic Cancer Organoid; FPCO)の創出に成功した。FPCOは膵癌を特徴づけているdesmoplastic reaction(間質線維化反応)や高い抗がん剤耐性を再現している。我々は、現在、FPCOを用いて、膵癌が示す免疫抑制性の制御機構の解明や膵癌微小環境をターゲットとした新規膵癌治療薬のスクリーニングなどを進めている。
Pancreatic ductal adenocarcinoma(PDAC) has a poor prognosis, with a 5-year survival rate of less than 10% due to delayed diagnosis, resistance to chemotherapy, and high metastatic potential. In addition, the tumor microenvironment(TME) of PDAC is characterized by the presence of complex stroma consisting of abundant extracellular matrix (ECM) and stromal cells such as cancer-associated fibroblasts (CAFs), tumor endothelial cells (TECs), and a variety of immune cells. This TME has been correlated with chemotherapy resistance and metastasis. Recently, we have generated a novel pancreatic cancer organoid (Fused pancreatic cancer organoids (FPCO) by co-culturing patient-derived pancreatic cancer cells with human iPS cell-derived mesenchymal and endothelial cells. Our FPCOs recapitulate TME, including desmoplastic reaction and resistance against anti-tumor drugs. We are investigating the immune-suppressiveness of PDACs and establishing a new drug screening system to identify a novel anti-tumor drug targeting TME.
ヒトiPS細胞を駆使した革新的な膵癌オルガノイドの創出 ---複雑で不均一な生体内癌微小環境の再現---
Incorporation of human iPSC-derived stromal cells creates a pancreatic cancer organoid with heterogeneous cancer-associated fibroblasts